Why Some Coatings Cause Water to Bead and Roll Off
Understanding the “blades of grass” effect in hydrophobic coating technology
Hydrophobic coatings are often described as coatings that make water bead, roll off or avoid wetting a surface. That description is useful, but it does not fully explain how the technology works.
The behaviour is controlled by a combination of surface chemistry and surface structure. A low-energy chemistry helps water avoid bonding strongly to the surface, while microscopic texture can reduce the real contact area between the water and the material beneath it.
A simple way to understand this is to imagine a water droplet sitting on the tips of blades of grass. The droplet is not fully touching the ground below. It is partly supported by the surface peaks, with air trapped underneath.

Structured hydrophobic surfaces reduce water contact area by allowing droplets to sit on microscopic surface peaks with air trapped underneath.
Hydrophobicity Is Not Just a Coating Material
Hydrophobic behaviour is not created by thickness alone. A very thin coating can be highly water repellent if the surface energy and surface interaction are controlled correctly.
In practical coating terms, this means that water repellency depends on how a droplet interacts with the outer surface of the coating, not simply how much coating has been applied.
Key point: hydrophobic performance is a surface effect. The outer few nanometres or microns often matter more than the total coating thickness.
The main factors are:
- Surface energy of the coating chemistry
- Surface roughness or microstructure
- Contact angle of the water droplet
- Sliding angle, or how easily the droplet moves
- Whether air can remain trapped under the droplet
- How contamination, oils or condensation affect the surface
The Blades of Grass Analogy
Imagine pouring water onto a perfectly flat surface. If the surface has high surface energy, the water spreads because it has good contact with the material beneath it.
Now imagine the same droplet resting on a dense field of grass. The droplet sits partly on the tips of the blades, with small pockets of air underneath. The water has less contact with the solid surface, so it is easier for the droplet to remain rounded and move away.
This is a useful analogy for structured hydrophobic surfaces. The surface may look smooth to the eye, but at a microscopic or nanoscopic level it can behave like a landscape of peaks, valleys and air pockets.
Flat surface versus structured surface
Smooth high-energy surface
Water spreads more easily because the droplet makes broad contact with the surface.
Smooth low-energy surface
Water beads more readily, but the droplet may still remain attached if contact area is high.
Structured hydrophobic surface
Water can sit on surface peaks, reducing contact area and improving roll-off behaviour.
Contact Angle and Sliding Angle
Contact angle describes the shape of a water droplet on a surface. A low contact angle means the water spreads. A high contact angle means the water beads.
However, contact angle does not tell the whole story. A droplet can have a high contact angle but still stick to the surface if adhesion is high. This is why sliding angle is also important.
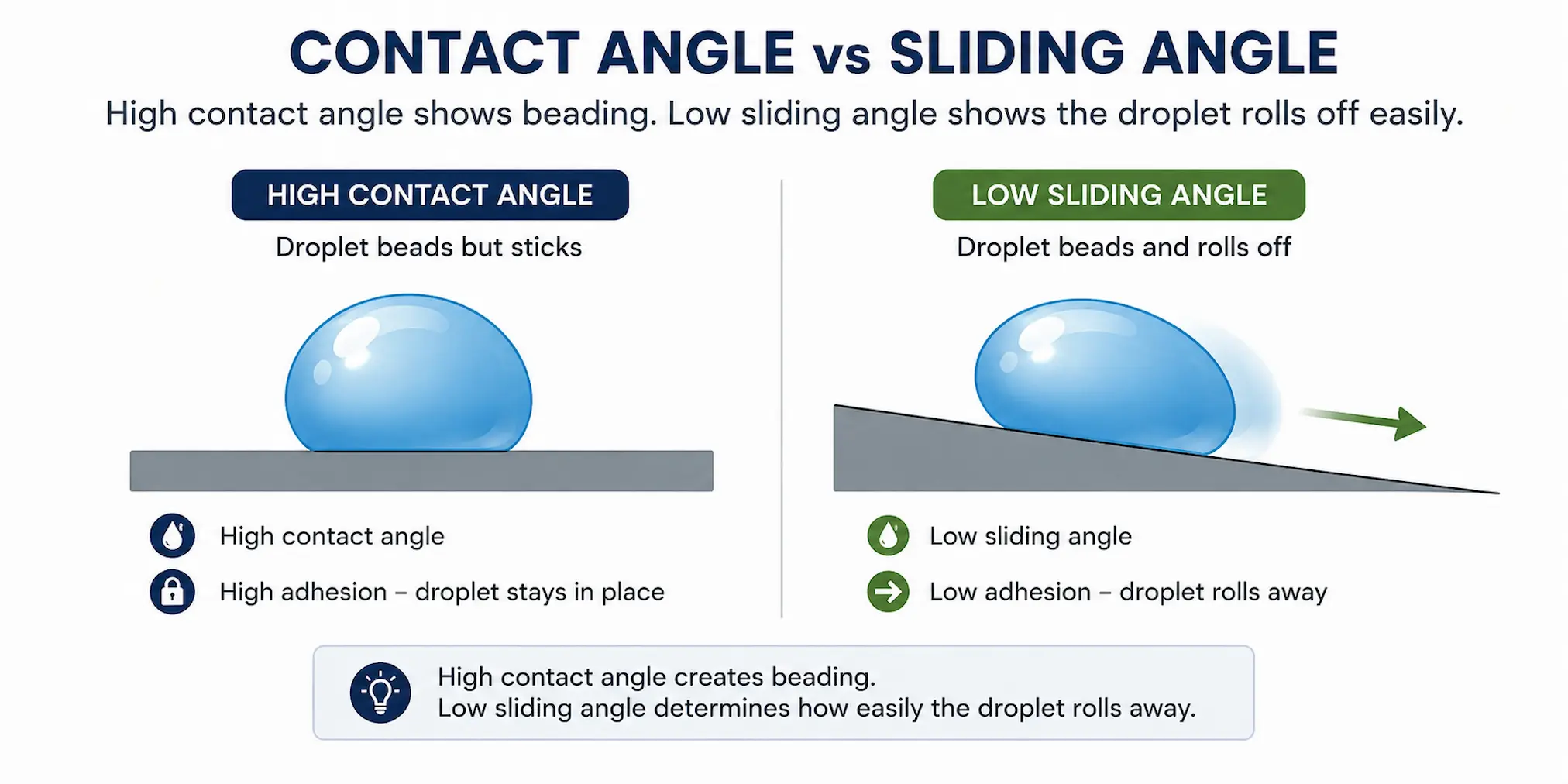
A high contact angle may create strong water beading, but low sliding angle determines how easily droplets roll away from the surface.
Why sliding behaviour matters
Sliding angle describes how easily a droplet moves when the surface is tilted. For practical hydrophobic coating performance, easy roll-off can be more important than appearance alone.
- High contact angle shows strong beading behaviour
- Low sliding angle shows easier droplet removal
- Low adhesion reduces water retention
- Reduced contact area supports self-cleaning behaviour
A coating that beads water but holds droplets in place may look hydrophobic, but it may not perform well where drainage, drying or contamination release are required.
Surface Energy: Why Chemistry Still Matters
The blades of grass analogy explains the structural part of hydrophobicity, but chemistry is still essential. The surface must discourage strong attraction between the water and the coating.
Fluoropolymer and other low-energy coating chemistries are often used because they reduce the ability of water to wet the surface.
Extremely low surface energy can also create process challenges in some coating systems, particularly where adhesion or wetting is required. This is one reason why de-wetting defects can occur on contaminated or low-energy surfaces.
In some electronics applications, plasma cleaning is used to increase surface energy and improve coating wetting and adhesion before conformal coating, Parylene deposition or ultra-thin coating application.
Structure and chemistry work together
The most effective hydrophobic surfaces often combine low-energy chemistry with controlled surface structure. The chemistry reduces attraction. The structure reduces contact area. Together, they help water remain mobile.
Low surface energy
Reduces the tendency of water to bond to or spread across the surface.
Surface texture
Reduces the real area of contact between the droplet and the coating.
Air entrapment
Allows the droplet to sit partly on air rather than fully wetting the solid surface.
Why Ultra-Thin Hydrophobic Coatings Can Be Effective
Ultra-thin hydrophobic coatings do not work like traditional protective conformal coatings. They are not designed primarily to create a thick physical barrier.
Instead, they modify the surface interaction between the substrate and the environment. This can make them useful where water repellency, reduced surface wetting or easier drying is required, but where a thicker coating may be unsuitable.
Where this matters
- Optical or sensor surfaces where coating thickness must be tightly controlled
- Assemblies where low mass or low build-up is required
- Components where water repellency is needed without traditional coating thickness
- Applications where the surface behaviour matters more than bulk coating protection
This is why hydrophobic coatings should not be judged only against conformal coatings by thickness. They often sit in a different functional category.
Hydrophobic Does Not Always Mean Protective
A hydrophobic coating can reduce water wetting, but that does not automatically mean it provides the same environmental protection as a conformal coating or Parylene coating.
For electronic assemblies, the difference is important. Water repellency may help reduce surface wetting, but long-term protection can also depend on coating continuity, thickness, edge coverage, ionic cleanliness, adhesion, mechanical durability and chemical resistance.
Hydrophobic behaviour helps manage water interaction. It should not be assumed to replace qualified environmental protection without testing.
Common misunderstanding
A surface can repel a water droplet in a simple demonstration but still fail in condensation, immersion, contamination, abrasion or chemical exposure. The test method must match the real operating environment.
How This Fits with Nano Coatings, Conformal Coatings and Parylene
Hydrophobic coatings sit within a wider coating selection landscape. They may be used as very thin surface-functional films, as advanced fluoropolymer coatings, or as part of a broader protection strategy.
They should be compared against other coating technologies by function, not just by thickness.
Nano coatings
Very thin surface-functional coatings that can provide water repellency with minimal build-up.
Hydrophobic coatings
Coatings selected primarily for water interaction, surface energy control and droplet behaviour.
Conformal coatings
Thicker protective coatings used to provide environmental protection for electronic assemblies.
Parylene coatings
Vapour-deposited polymer coatings used where highly uniform coverage and barrier performance are required.
For further comparison, see our articles on nano coating vs conformal coating, when to use hydrophobic coatings and why conformal coating fails on complex PCB assemblies.
Practical Selection Questions
Before selecting a hydrophobic coating, it is important to define what the coating is expected to do. Water beading alone is not a complete specification.
Useful questions include:
- Is the coating required to repel droplets, resist condensation or survive immersion?
- Is the main requirement water repellency, chemical resistance or electrical protection?
- Will the surface be touched, rubbed, cleaned or abraded?
- Does the coating need to remain optically clear?
- Are connectors, vias, test points or contact areas present?
- Is thickness build-up acceptable?
- How will coating performance be verified after application?
These questions help separate a surface-functional coating requirement from a true environmental protection requirement.
Related Technical Guidance
The following pages provide additional context for selecting and comparing coating technologies:
- Advanced Functional Coatings – overview of surface-functional coating technologies.
- Hydrophobic Coatings – commercial coating pathway for water-repellent surface behaviour.
- Ultra-Thin Coatings – coating options where low build-up and surface function are important.
- Nano Coating PCB Limitations – practical limits of very thin coating systems.
Why Choose SCH Services?
SCH Services supports customers with coating selection, process development, coating application and technical troubleshooting across conformal coating, Parylene coating and advanced functional coating technologies.
- Advanced Functional Coatings for hydrophobic, ultra-thin and specialist surface-functional requirements.
- Conformal Coating Solutions for electronics protection, process support and production coating requirements.
- Parylene Training and Support for process understanding, masking control and production implementation.
Where coating choice is uncertain, SCH can help review the application, define the functional requirement and identify which coating technology is most appropriate for validation.
Summary
The “blades of grass” analogy helps explain why hydrophobic coatings can be effective even when they are very thin. Water repellency is controlled by how a droplet interacts with the surface, not simply by coating thickness.
Low surface energy helps water bead. Surface structure can reduce contact area. Air trapped beneath the droplet can make it easier for water to roll away.
However, hydrophobic behaviour should not be confused with complete environmental protection. The coating must be selected and tested against the real exposure conditions, including condensation, contamination, abrasion, chemicals and electrical reliability requirements.
Disclaimer: This article is provided as general technical guidance only. Hydrophobic coating selection, coating performance and protection claims must be validated against the specific substrate, operating environment, test method, customer specification and applicable industry standards. Final coating decisions should be confirmed through appropriate qualification testing.

